LPG Storage Requirements - LPG Storage Regulations NZ - LPG Tank Safety - Bulk Gas Storage LPG gas storage requirements (bulk gas storage)...
ELGAS Knowledge Hub
Gas Bottle Disposal – Gas Bottle Recycling – Where to Dispose of Gas Bottles Near Me – Community Recycling Centre
Gas Bottle Disposal - Gas Bottle Removal - Gas Bottle Recycling - Where to Dispose of Gas Bottles Near Me Free - Community Recycling...
How to Check Gas Bottle Level: Gas Bottle Level Indicator – Gas Bottle Gauge
How to Check Gas Bottle Level: Gas Bottle Level Indicator - Gas Bottle Gauge How to check gas bottle level using a gas bottle level...
Mega Joules: KJ to Megajoules – MJ to KJ – MJ to kWh – KJ to MJ – MJ to Joules – Megajoules to Kilowatts
Mega Joules: KJ to Megajoules, MJ to KJ, Megajoules to Kilowatts, KJ to MJ MJ to kWh, MJ to Joules, Megajoules to Kilowatts (MJ kW),...
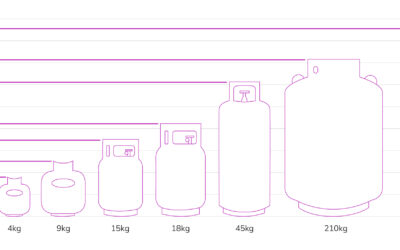
LPG Gas Bottle Sizes NZ – Gas Cylinders – Gas Tanks – LPG Gas Bottle Dimensions
LPG Gas Bottle Sizes NZ - Gas Cylinders - LPG Gas Tanks - LPG Gas Bottle Dimensions - Smallest Gas Bottle This guide to LPG gas bottles...
LPG FAQs
How does LPG work? LPG – Liquefied Petroleum Gas – is stored under pressure as a liquid in a gas bottle. It turns back into a gas when you...
Gas Bottle Clearances NZ – LPG Bottle Clearances – Gas Bottle Location Regulations NZ
Gas Bottle Clearances NZ - LPG Bottle Clearances - Gas Bottle Location Regulations NZ Gas bottle installation regulations NZ (gas bottle...
Liquified Petroleum Gas, LP Gas, Liquefied Petroleum Gas LPG – LPG vs Natural Gas – What is LPG
Liquified Petroleum Gas, LP Gas, Liquefied Petroleum Gas LPG - LPG vs Natural Gas - LPG in Gas Liquified petroleum gas (liquid petroleum...
Gas Bottle Changeover Switch – Gas Changeover Valve NZ – 45kg Gas Bottle Regulator
Gas Bottle Changeover Switch - Gas Changeover Valve NZ - 45kg Gas Bottle Regulator New to Home LPG? You Are In The Right Place! Learn...